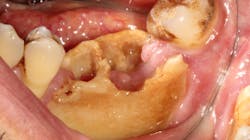
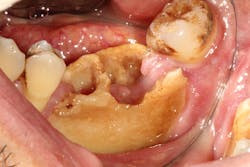
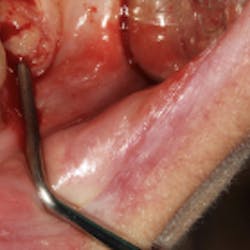
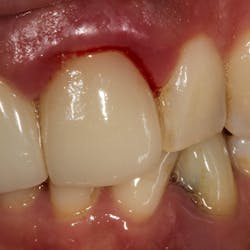
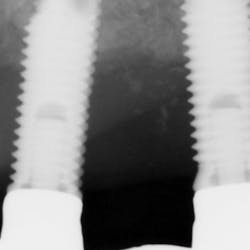
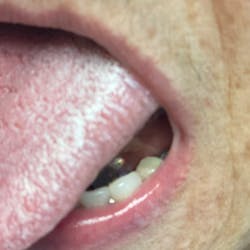
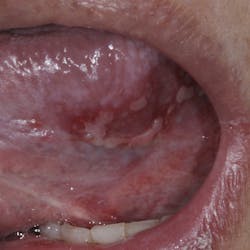
Although most rheumatologists would prefer to prescribe the historically tested BPs with their strong track record and known risk-benefit analysis, there are alternative medications to treat osteoporosis that appear to be associated with less risk of MRONJ. Little is known, however, about the long-term safety of these medications. I have been practicing long enough to remember when NSAIDs were new medications. Soon after they became available, I consulted with an obstetrician regarding what pain medication to use for a pregnant woman who had a painful abscess. My thought was that using an NSAID would be safer to the fetus than a narcotic, and I suggested this to my medical colleague. The physician, however, said that Tylenol #3 (acetaminophen/codeine) had been safely used for many years in pregnant women, but the same could not be said for ibuprofen.
ADDITIONAL READING ...
Antidepressant medications and nutraceuticals: Effects on the success of dental implants
Medication-induced bleeding when your patient 'doesn't take any medication'
What follows is a general review of alternative drugs used to treat osteoporosis that have little or no known association with MRONJ.
Denosumab (Prolia)
The antiresorptive agent denosumab is a fully humanized monoclonal antibody against RANKL (receptor activator of nuclear factor-kappa B), which is administered subcutaneously. RANK is a surface receptor of preosteoclasts and is activated by RANKL (the RANK-Ligand), which is found on the surface of osteoblasts (bone-forming cells). Activation of RANK by RANKL promotes the maturation of preosteoclasts into osteoclasts (bone degradation cells). Denosumab inhibits the maturation of osteoclasts by binding to and inhibiting RANKL. This mimics the RANKL action of osteoprotegerin, an endogenous substance theorized to be present in decreased concentrations in patients with osteoporosis. Both denosumab and osteoprotegerin protect the bone from degradation and prevent progression of the disease. The risk for MRONJ is 0.04%, or four cases per 10,000 patients exposed to denosumab. The good news with this medication is that six months after administration there is none remaining in the bone, and dental surgery can be safely accomplished. Arranging a “drug holiday” with the rheumatologist can provide you with a safe window to complete elective surgery for your patients. If my patient opts for this drug, I will complete all surgery prior to her beginning Prolia and hopefully eliminate any MRONJ risk. If problems arise in the future, a drug holiday will give us a safe time frame during which she can be treated. You can find out more about how Prolia works at this link.
Teriparatide (Forteo)
Teriparatide is a recombinant form of the active 34 amino acid N-terminus of parathyroid hormone (PTH). As does PTH, teriparatide activates osteoblasts more so than osteoclasts, thus improving bone density. There appears to be no etiologic association of MRONJ with teriparatide. In fact, it has been used successfully to treat MRONJ. In addition to increasing bone density, other medical uses include treating the nonunion of bone fractures. Apparently it has been used off-label in professional athletes to speed the healing of bone fractures. You can find out more about Forteo at this link.
Raloxifene (Evista)
The drug raloxifene’s biological actions are mediated through binding to estrogen receptors. In some tissues this binding results in activation of estrogenic pathways (agonism), and other tissues have their estrogenic pathways blocked (antagonism). The antagonistic/agonistic stimulation of certain receptors has earned this drug a place in the class of selective estrogen receptor modulators (SERM). Raloxifene acts as an estrogen agonist in bone, thus continuing to maintain bone density as menopause reduces endogenous estrogen. Both raloxifene and its older relative, tamoxifene, are useful in the treatment of estrogen-receptor-positive breast cancer. Raloxifene, perhaps due to fewer side effects, has become more widely used in osteoporosis therapy than tamoxifene. Raloxifene has no known association with MRONJ.