A new minimally invasive bone grafting technique: SMART
The subperiosteal minimally invasive esthetic ridge augmentation technique called SMART—developed as an alternative to traditional guided bone regeneration procedures—has the potential to profoundly impact the future of bone augmentation. Ernesto A. Lee, DMD, explains more about the clinical features of this new bone grafting technique.
Traditional guided bone regeneration (GBR) techniques include flap mobilization and placement of a bone graft, often with the use of space-maintaining devices and cell occlusive membranes. This approach is frequently associated with complications, such as incomplete wound closure and soft-tissue dehiscence that may lead to exposure and infection of the membrane and graft material. (1) These complications may not only jeopardize the outcome of the bone augmentation but also result in increased morbidity, including pain, swelling, infection, and delayed healing.
The effects of these procedures on the peri-implant soft-tissue architecture, and the potential esthetic sequelae are important considerations. There is growing evidence that bone augmentation and guided bone regeneration procedures frequently result in soft-tissue defects. (2,3)
The subperiosteal minimally invasive esthetic ridge augmentation technique (SMART) was developed as an alternative to the treatment of complex, high-esthetic-risk scenarios, where any gingival disfigurement secondary to traditional GBR procedures was not acceptable.
The technique uses specially designed instruments and a laparoscopic approach to deliver a growth factor/xenograft combination into a previously created subperiosteal pouch. Flap elevation, membranes, tenting screws, and decortication are not required.
A recent publication in the International Journal of Periodontics and Restorative Dentistry reported the results of a prospective case series where the SMART procedure was used to treat 60 sites on 21 patients with a follow-up period ranging from four to 30 months. Five treatment categories were included, with 25 implants placed and 21 restored. Additionally, the article presents the results from human histologic sections. (4)
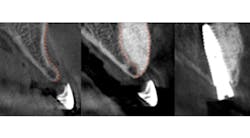
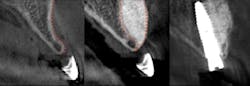
Predictable bone regeneration was observed and all dental implants were successful, with no evidence of crestal bone loss (figure 1). Only one postoperative complication was reported in which particulate granules were exfoliated through a residual fistulous communication. A survey of patient-based outcomes revealed that morbidity and complication rates were reduced as well.
Figure 1: Horizontal augmentation of patient with congenitally missing lateral incisor. Implant was placed four months following SMART bone graft.
The average horizontal augmentation achieved with the SMART method for all treatment categories was 5.11 mm (SD=0.76). Clearly, a greater increase in horizontal ridge width is to be expected following augmentation of an edentulous ridge than when grafting bone lateral to an existing tooth or implant.
The mean horizontal augmentation of edentulous ridges using the SMART method was 6.47 mm (SD=1.40). This compares favorably with outcomes reported by Urban and coworkers in 2013, Proussaefs and Lozada in 2006, and Pieri et al in 2008. (5–7)
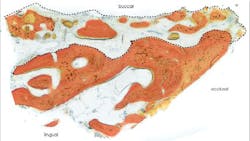
Human histology results demonstrated xenograft particles surrounded by newly formed bone. Histomorphometric measurements revealed a 52% bone content in the sample (figure 2).
Figure 2: Histologic section of core sample harvested during implant osteotomy preparation.
Since no decortication or intramarrow penetration was performed in the current case series, the role of the periosteum as a source of pluripotential cells in growth factor-mediated bone regeneration must be considered. (8–11)
Although the incidence of complications is significantly less with SMART than with traditional GBR, it is nevertheless a technique-sensitive procedure. The need for training and the use of specially designed instrumentation and adequate biomaterials cannot be overemphasized. However, with better outcomes, no flaps, no membranes or tenting screws, and fewer complications, SMART has the potential to profoundly impact the future of bone augmentation.
Editor’s note: Dr. Ernesto Lee will be introducing this novel minimally invasive technique at the Academy of Osseointegration's Annual Meeting in Orlando, Florida, during presentations in the Corporate Forum, Managing Anterior Esthetics, and Clinical Innovations sections of the program. Visit the AO website for more information about the 2017 Annual Meeting, March 15–18.
Author’s note: To learn more about SMART or to register for the June 23–24 training and certification course, visit drernestolee.com or contact [email protected].
References
1. Jensen AT, Jensen SS, Worsaae N. Complications related to bone augmentation procedures of localized defects in the alveolar ridge. A retrospective clinical study. Oral Maxillofac Surg. 2016;20(2):115-122. doi: 10.1007/s10006-016-0551-8.
2. Cosyn J, Eghbali A, Hanselaer L, et al. Four modalities of single implant treatment in the anterior maxilla: a clinical, radiographic, and aesthetic evaluation. Clin Implant Dent Relat Res. 2013;15(4):517-530. doi: 10.1111/j.1708-8208.2011.00417.x.
3. Cosyn J, Sabzevar MM, De Bruyn H. Predictors of interproximal and midfacial recession following single implant treatment in the anterior maxilla: a multivariate analysis. J Clin Periodontol. 2012;39(9):895-903. doi: 10.1111/j.1600-051X.2012.01921.x.
4. Lee EA. Subperiosteal minimally invasive aesthetic ridge augmentation technique (SMART): A new standard for bone reconstruction of the jaws. Int J Periodontics Restorative Dent. 2017;37(2):165-163. doi: 10.11607/prd.3171.
5. Urban IA, Nagursky H, Lozada JL, Nagy K. Horizontal ridge augmentation with a collagen membrane and a combination of particulated autogenous bone and anorganic bovine bone-derived mineral: a prospective case series in 25 patients. Int J Periodontics Restorative Dent. 2013;33(3):299-307. doi: 10.11607/prd.1407.
6. Proussaefs P, Lozada J. Use of titanium mesh for staged localized alveolar ridge augmentation: clinical and histologic-histomorphometric evaluation. J Oral Implantol. 2006;32(5):237-247.
7. Pieri F, Corinaldesi G, Fini M, Aldini NN, Giardino R, Marchetti C. Alveolar ridge augmentation with titanium mesh and a combination of autogenous bone and anorganic bovine bone: a 2-year prospective study. J Periodontol. 2008;79(11):2093-2103. doi: 10.1902/jop.2008.080061 .
8. Malizos KN, Papatheodorou LK. The healing potential of the periosteum molecular aspects. Injury. 2005;36(Suppl 3):13-19.
9. Roberts SJ, Geris L, Kerckhofs G, Desmet E, Schrooten J, Luyten FP. The combined bone forming capacity of human periosteal derived cells and calcium phosphates. Biomaterials. 2011;32(19):4393-4405. doi: 10.1016/j.biomaterials.2011.02.047.
10. Zhu SJ, Choi BH, Huh JY, Jung JH, Kim BY, Lee SH. A comparative qualitative histological analysis of tissue-engineered bone using bone marrow mesenchymal stem cells, alveolar bone cells, and periosteal cells. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101(2):164-169.
11. Ceccarelli G, Graziano A, Benedetti L, et al. Osteogenic potential of human oral-periosteal cells (PCs) isolated from different oral origin: an in vitro study. J Cell Physiol. 2016;231(3):607-612. doi: 10.1002/jcp.25104.