A new pill to treat sleep apnea
Sleep apnea—especially obstructive sleep apnea (OSA)—is a rising health concern, with up to 80% of people remaining undiagnosed. Sleep apnea affects 30–40 million people in the United States and 1 billion people worldwide.1
Types of sleep apnea
There are two types of sleep apnea2:
- Central sleep apnea occurs when the muscles and nerves aren’t activated enough for breathing while asleep, causing pauses in airflow. Obstructive sleep apnea occurs when the upper airway becomes blocked during sleep, interfering with breathing.
Diagnosis
Based on your symptoms and a number called the apnea-hypopnea index (AHI) found during a sleep study, you may be diagnosed with mild (5–14 episodes), moderate (15–30 episodes), or severe (30-plus episodes) OSA. The AHI measures the number of times your breathing was stopped or hindered as you slept.
- Risk factors: male, older, smoking, asthma, thick neck, smaller airway, large tongue, large tonsils, nonwhite, family history of sleep apnea
- Health risk factors: obesity, asthma, neurological disorders, hypothyroidism, cleft lip/palate, heart disease, kidney disease, and polycystic ovary syndrome (PCOS)
- Signs and symptoms: loud snoring, sore throat in morning, gasping for air during sleep, night sweats, daytime sleepiness, dry mouth, headaches (especially in morning), frequent waking or waking to urinate, high blood pressure, and gastroesophageal reflux disorder (GERD) Treatments: continuous positive airway pressure (CPAP) machines, oral appliances, upper airway stimulation, surgery, maxillary/mandibular advancement, somnoplasty. More common forms of treatment for moderate to severe OSA include CPAP machines, which require a mask that forces air through the nose/mouth and keeps the upper airway from relaxing and closing off (figure 1). Compliance, discomfort, and disturbance to sleep partners are more of the common complaints.
A new pill to treat sleep apnea
A new pill called AD109, currently in phase III clinical trials, has been created to relieve the symptoms of sleep apnea and prevent the upper airway from closing.3 AD109 is a combination of the drugs aroxybutynin and atomoxetine. Aroxybutynin is used to treat symptoms of an overactive bladder, while atomoxetine is used to treat attention deficit hyperactivity disorder.
For phase II trials of AD109, patients were separated into groups based on their AHI indices. Besides reporting no serious adverse side effects, one promising result out of the phase II trials was the lack of major side effects and marked improvements in chronic fatigue. The second promising result in people who took the drug—those with an index score of 10–15 (mild)—77% had their scores lowered to below 10. Forty-two percent with a score of 15–30 (moderate) were able to get below 10. Seven percent of those with a score over 30 were able to get to 10 or below.
Limitations of this study
More research is needed to validate the use of this new drug treatment. Limitations of the study include the omission of blood oxygen saturation improvements in patients taking this medication. Instead, hypoxic burden was described. With sleep apnea, the body takes in less oxygen during sleep, which causes many of its effects when you’re awake.
One critic of the study (Douglas Kirsch, MD, medical director of sleep medicine at Atrium Health in Charlotte, NC) suggests: “There's a big difference between somebody who has a minimum oxygen saturation of 89% and went from an AHI of 20 to 12 … which sounds great … but had minimum oxygen saturation stay the same after. If 99% of a sleep study was at 90% and above, but there was one dip at 80%, that’s not the same as spending 45 minutes below 88%. What you really want to talk about is how much or how long does that oxygen get low?”
Limitations of “just a pill” concept to treat sleep apnea
According to coauthor Dr. Erin Elliott, “These drugs can really work as they help with upper airway dilation, and they need this drug combination because muscles are different in REM sleep versus non-REM sleep. The problem is that the medical world works too much in silos or on islands. Dentistry and medicine have to work together.”
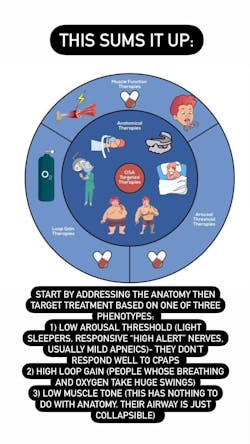
CPAP machines, oral appliances, surgery, weight loss, etc., help treat the anatomical issue, which is one part of the cause. The other cause is one of three categories: low-arousal threshold (light sleepers, mild patients), high loop gain (obese people in whom oxygen desaturates severely because their breathing receptors are not working correctly), and people with low muscle tone (figure 2).
“So, these drugs in combination with an appliance would be awesome. We must start tailoring treatments based on phenotypes and not a one-treatment-fits-all philosophy.”
This study does, however, offer promise in advancing OSA treatment therapeutics, especially for the treatment of patients who suffer from mild to moderate forms of sleep apnea.
Editor’s note: This article originally appeared in Perio-Implant Advisory, a chairside resource for dentists and hygienists that focuses on periodontal- and implant-related issues. Read more articles and subscribe to the newsletter.
References
- Ling V. Sleep apnea statistics and facts you should know. National Council on Aging. May 8, 2024. https://www.ncoa.org/adviser/sleep/sleep-apnea-statistics/
- What is sleep apnea? National Heart, Lung, and Blood Institute. Updated March 24, 2022. https://www.nhlbi.nih.gov/health/sleep-apnea
- Parallel-arm study to compare AD109 and placebo with patients with OSA (SynAIRgy Study). ClinicalTrials.gov. U.S. National Library of Medicine. April 14, 2023. Updated June 3, 2024. https://classic.clinicaltrials.gov/ct2/show/NCT05813275
About the Author

Scott Froum, DDS
Editorial Director
Scott Froum, DDS, a graduate of the State University of New York, Stony Brook School of Dental Medicine, is a periodontist in private practice at 1110 2nd Avenue, Suite 305, New York City, New York. He is the editorial director of Perio-Implant Advisory and serves on the editorial advisory board of Dental Economics. Dr. Froum, a diplomate of both the American Academy of Periodontology and the American Academy of Osseointegration, is in the fellowship program at the American Academy of Anti-aging Medicine, and is a volunteer professor in the postgraduate periodontal program at SUNY Stony Brook School of Dental Medicine. He is a trained naturopath and is the scientific director of Meraki Integrative Functional Wellness Center. Contact him through his website at drscottfroum.com or (212) 751-8530.
Erin Elliott, DDS
Erin Elliott, DDS, grew up in southern California but went away to a small NAIA school in western New York where she played collegiate soccer and graduated summa cum laude from Houghton College. After graduating from Creighton Dental School in 2003, she settled in north Idaho to begin her general dentistry career. She has a special interest in dental sleep medicine and has lectured extensively on this topic. Dr. Elliott is an active member of various dental associations, including the American Academy of Sleep Medicine and American Academy of Dental Sleep Medicine. She is the past president and a diplomate of the American Sleep and Breathing Academy. She teaches a two-day sleep apnea implementation course with 3D Dentists and Tarun Agarwal, DDS.