P-tau blood biomarkers in the dental chair: Why p-tau positivity should change periodontal management
Key Highlights
- Plasma p-tau181 and p-tau217 are emerging as highly sensitive early biomarkers for Alzheimer’s disease (AD), mild cognitive impairment (MCI), and broader neurodegenerative processes.
- Biomarker positivity often precedes clinical symptoms, creating a critical window for early intervention.
- Periodontal disease is a modifiable inflammatory risk factor linked to neurodegeneration through systemic and microbial pathways.
- Dental professionals are uniquely positioned to identify, monitor, and intervene in at-risk patients.
- Patients testing positive for p-tau biomarkers should be considered for intensified periodontal surveillance and treatment protocols.
The conversation around Alzheimer’s disease and neurologic degeneration has shifted dramatically in recent years. We are no longer limited to diagnosing disease after cognitive decline has occurred. Instead, we are now identifying biologic signals of neurodegeneration years—if not decades—before symptoms emerge. Among the most promising of these signals are plasma phosphorylated tau biomarkers—specifically p-tau181 and p-tau217. For periodontists and dental clinicians, this is more than a neurologic breakthrough—it is a call to action.
P-tau biomarkers: Detecting disease before it manifests
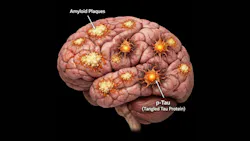
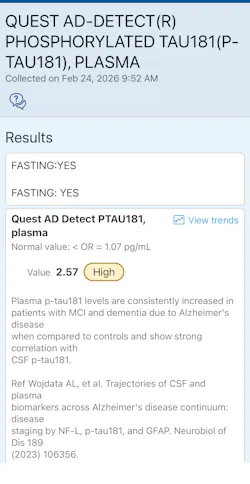
Phosphorylated tau proteins reflect abnormal tau processing and aggregation—hallmarks of neurocognitive decline and Alzheimer’s pathology. What makes p-tau181 and p-tau217 particularly compelling is their detectability in plasma, allowing for scalable, minimally invasive screening (figures 1 and 2).
In a pivotal study, plasma p-tau181 was shown to predict both cognitive and functional decline, even in individuals without overt dementia at baseline. The authors concluded that “Plasma p-tau181 is associated with longitudinal cognitive decline and may serve as an early biomarker of disease progression.”1
This positions p-tau181 not just as a diagnostic adjunct, but as a prognostic tool—identifying patients on a trajectory toward impairment.
Similarly, emerging evidence suggests that p-tau217 may be even more tightly linked to amyloid pathology, one of the earliest drivers of Alzheimer’s disease. A 2026 meta-analysis by Malek-Ahmadi et al. demonstrated that: “Plasma p-tau217 correlates strongly with amyloid burden, even in cognitively unimpaired individuals.”2
In practical terms, this means patients may test positive for p-tau217 years before any clinical signs of MCI or Alzheimer’s disease appear. Further reinforcing this, one study identified elevated Alzheimer’s-related biomarkers in patients with neuropsychiatric symptoms, suggesting that neurodegenerative processes may be underway even in atypical or early presentations.3
Another study extended these findings by highlighting sex-based differences in p-tau217 expression, tau aggregation, and cognitive decline, emphasizing the need for personalized risk assessment.4
How p-tau testing is relevant to dentistry
At first glance, tau pathology and periodontal disease may seem unrelated. But mounting evidence tells a different story. Chronic periodontitis is not confined to the oral cavity. It is a systemic inflammatory condition with well-documented effects on cardiovascular disease, diabetes—and increasingly—neurodegeneration.5
The link is multifactorial:
- Systemic inflammation (IL-1β, TNF-α, CRP)
- Bacterial dissemination, particularly Porphyromonas gingivalis
- Neuroinflammatory signaling pathways
- Blood–brain barrier disruption
Periodontitis and P. gingivalis activate inflammatory pathways that may contribute directly to neurodegenerative changes and Alzheimer’s disease progression.6 This is not theoretical. Studies have identified oral pathogens and their virulence factors in brain tissue, suggesting a direct microbial contribution to disease.
The clinical intersection: Biomarker positivity meets modifiable risk
If p-tau181 and p-tau217 identify patients at increased risk for neurodegeneration, and periodontal disease contributes to systemic inflammation and possibly amyloid/tau pathology, then periodontal therapy becomes more than oral care—it becomes risk modification. This creates a new category of dental patient:
The “neurologically at-risk” periodontal patient
Patients who:
- Test positive for p-tau181 or p-tau217
- Have a family history of Alzheimer’s disease
- Apo E4/E4 positive
- Exhibit early cognitive or neuropsychiatric changes
- Present with moderate to severe periodontal inflammation
These individuals should not be managed with routine recall alone.
Proposed clinical approach in the dental office
No. 1: Risk stratification
Patients reporting:
- Neurologic screening tests (p-tau biomarkers)
- Memory concerns or family history
→ Should be flagged for enhanced periodontal evaluation
No. 2: Aggressive inflammation control
This includes:
- Scaling and root planing
- Local antimicrobial therapy
- Host modulation (where appropriate)
- Surgical intervention when indicated
The objective is not just pocket reduction—it is systemic inflammatory burden reduction.
No. 3: Shortened recall intervals
For p-tau–positive patients:
- Three-month periodontal maintenance (minimum)
- Consider two-month intervals in high-risk cases
No. 4: Interdisciplinary collaboration
Communication with:
- Neurologists
- Primary care physicians
- Functional medicine providers and naturopathic doctors
Can periodontal treatment influence neurodegenerative risk?
There is no direct evidence yet that treating periodontal disease prevents Alzheimer’s disease. There is, however, moderate to strong data showing that periodontal treatment reduces Alzheimer’s disease imaging markers.7
For example, there is strong evidence that:
- Systemic inflammation accelerates neurodegeneration.
- Periodontal disease is a major contributor to systemic inflammation.
- Periodontal therapy reduces inflammatory markers.
This creates a biologically plausible and clinically actionable pathway. In other words: We may not yet be able to cure Alzheimer’s, but we can potentially reduce one of its modifiable accelerants.
A shift in perspective: Dentistry as preventive neurology
Dentistry has historically been siloed from medicine. That model is no longer sustainable.
If we accept that:
- p-tau biomarkers identify early disease susceptibility, and
- periodontal inflammation contributes to systemic and neurologic burden,
then the dental office becomes a frontline screening and intervention point. This is particularly relevant because patients see dentists more frequently than physicians—often twice per year or more. That frequency creates opportunity.
Conclusion
The emergence of p-tau181 and p-tau217 as early biomarkers of neurodegeneration represents a major advance in medicine. Biomarkers alone do not change outcomes—intervention does. Periodontal disease is one of the few modifiable, measurable, and treatable inflammatory conditions that may influence systemic health, including neurologic disease.
As clinicians, we should begin asking:
- Has this patient undergone biomarker screening?
- Are they at elevated neurologic risk?
- What can we do—today—to reduce their inflammatory burden?
The future of periodontology is not just about placing implants and saving teeth; it is about creating a customized treatment plan that protects the patient as a whole.
Editor’s note: This article originally appeared in Perio-Implant Advisory, a chairside resource for dentists and hygienists that focuses on periodontal- and implant-related issues. Read more articles and subscribe to the newsletter.
References
- Tropea TF, Waligorska T, Xie SX, et al. Plasma phosphorylated tau181 predicts cognitive and functional decline. Ann Clin Transl Neurol. 2023;10(1):18-31. doi:10.1002/acn3.51695
- Malek-Ahmadi M, Sharma S, Stipho F, et al. Plasma phosphorylated tau 217 and amyloid burden in older adults without cognitive impairment: a meta-analysis. JAMA Neurol. 2026;83(1):13-19. doi:10.1001/jamaneurol.2025.4721
- Bray MJC, Shaw JS, Morrow CB, Onyike CU, Alzheimer’s Disease Neuroimaging Initiative. Alzheimer disease-relevant biomarker elevations in psychosis and broad neuropsychiatric impairment. JAMA Psychiatry. 2026;83(3):318-320. doi:10.1001/jamapsychiatry.2025.4347
- Coughlan GT, Ourry V, Townsend D, et al. Sex differences in p-tau217, tau aggregation, and cognitive decline. JAMA Neurol. 2026:e255670. doi:10.1001/jamaneurol.2025.5670
- Tattar R, da Costa BDC, Neves VCM. The interrelationship between periodontal disease and systemic health. Br Dent J.2025;239(2):103-108. doi:10.1038/s41415-025-8642-2
- Froum S. Periodontitis, Porphyromonas gingivalis, and the inflammatory pathways linking gum disease to neurodegeneration. Perio-Implant Advisory. January 5, 2026. https://www.perioimplantadvisory.com/clinical-tips/article/55341395/periodontitis-porphyromonas-gingivalis-and-the-inflammatory-pathways-linking-gum-disease-to-neurodegeneration
- Schwahn C, Frenzel S, Holtfreter B, et al. Effect of periodontal treatment on preclinical Alzheimer's disease—results of a trial emulation approach. Alzheimer's Dement. 2022;18(1):127-141. doi:10.1002/alz.12378
About the Author

Scott Froum, DDS
Editorial Director
Scott Froum, DDS, a graduate of the State University of New York, Stony Brook School of Dental Medicine, is a periodontist in private practice at 1110 2nd Avenue, Suite 305, New York City, New York. He is the editorial director of Perio-Implant Advisory and serves on the editorial advisory board of Dental Economics. Dr. Froum, a diplomate of both the American Academy of Periodontology and the American Academy of Osseointegration, is in the fellowship program at the American Academy of Anti-aging Medicine, and is a volunteer professor in the postgraduate periodontal program at SUNY Stony Brook School of Dental Medicine. He is a trained naturopath and is the scientific director of Meraki Integrative Functional Wellness Center. Contact him through his website at drscottfroum.com or (212) 751-8530.

Micaela Milano, MBA
Micaela Milano, MBA, is the owner and founder of Meraki Integrative, where she combines her extensive professional experience and passion for holistic health to deliver personalized, transformative care. Micaela holds an MBA in business, marketing, and management from Florida International University. In 2018, she founded the Meraki Initiative, dedicating herself to integrative wellness and client empowerment. A certified 200-hour yoga teacher, Reiki practitioner, and advanced holistic health coach, Micaela also holds certifications in angelic and crystal healing, bringing a deeply personal and spiritual dimension to her work. Her approach centers on addressing the root causes of health concerns, merging functional medicine with regenerative esthetics to support both inner vitality and outer radiance.