Use of horizontal centrifugation of platelet-rich fibrin leads to a 4-fold increase in cell concentration
Key Highlights
- PRF enhances healing naturally: This autologous fibrin matrix promotes angiogenesis, cell recruitment, and sustained growth factor release—supporting faster, more effective tissue regeneration than earlier platelet concentrates like PRP.
- Evolution improves outcomes: Eliminating anticoagulants and adopting horizontal centrifugation increases cell content and healing potential, overcoming key limitations of traditional PRP methods.
- Wide clinical applications: PRF is used across dentistry for extraction sites, bone augmentation, implant procedures, periodontal regeneration, and soft tissue healing, with growing evidence of improved clinical results.
Abstract
Platelet-rich fibrin (PRF) has been utilized in implant dentistry for well over a decade owing to its ability to rapidly stimulate tissue angiogenesis and faster wound healing and recovery. Over the years, much progress has been made to more effectively concentrate platelets and growth factors as well as define their clinical use for the regeneration of both soft and hard tissues.
This overview article gives a brief history of platelet concentrates regarding the advancements made over the years from platelet-rich plasma (PRP) toward a more autologous three-dimensional scaffold found in PRF. Then, a deep understanding of the years of research leading toward appropriate protocols and methods to best concentrate PRF will be discussed using horizontal centrifugation. Lastly, clinical indications and uses of PRF will be demonstrated with case examples highlighting the use of PRF in extraction site management, bone augmentation, as well as in implant dentistry.
Introduction
Platelet concentrates have been utilized in medicine for over three decades owing to their ability to rapidly secrete autologous growth factors and ultimately speed wound healing.1 They have gained tremendous momentum as a regenerative agent derived from autologous sources capable of stimulating tissue regeneration in a number of medical fields.2-4 Many years ago, it was proposed that by concentrating platelets utilizing a centrifugation device, growth factors derived from blood could be collected from a PRP layer, and later utilized in surgical sites to promote local wound healing.3,4 Today, it has been well established that platelet concentrates act as a potent mitogen capable of:
- Speeding the revascularization of tissues (angiogenesis)
- Acting as a potent recruitment agent of various cells including stem cells
- Inducing the prompt multiplication of various cell types found in the human body (proliferation)
Specifically in dentistry, platelet concentrates were introduced over 20 years ago by Marx and colleagues with the aim of concentrating blood proteins as a natural source of growth factors that could stimulate vascularization (angiogenesis) and tissue ingrowth based on the fact that blood supply is pivotal for tissue regeneration of all tissues.5 Wound healing has been described as a four-step process which includes 1) hemostasis, 2) inflammation, 3) proliferation, and 4) maturation.6-8
The evolution of platelet-rich fibrin
It is interesting to point out that the use of platelet concentrates has slowly and gradually gained in popularity, with a dramatic increase being observed in the past five to 10 years (QR code 1). PRP, as its name implies, was designed to accumulate platelets in supraphysiological doses within the plasma layer following centrifugation. The main aim of PRP was to isolate and further concentrate the highest quantity of platelets and their associated growth factors for regenerative purposes and thereafter reimplanting this specialized supraconcentrate at sites of local injury.2
Initial protocols typically ranged in duration from 30 minutes to one hour based on the centrifugation/collection systems and protocols utilized. Since lengthy protocols were utilized, anticoagulants were added to blood collection tubes. These typically were either various forms/concentrated bovine thrombin and calcium chloride.
Despite its growing success and continued use, several reported limitations existed with respect to reaching its full healing potential. Mainly, the use of anticoagulants was shown to limit wound healing.2 Simply put, when injury occurs leading to an open wound, a blood clot is one of the first and most crucial steps that occurs in order for healing to take place. Shortly thereafter, cells and growth factors get trapped within this newly formed extracellular matrix, and the wound healing process/cascade begins. By limiting the body’s ability to form a clot-stable tissue, wound healing is delayed. Several studies have now demonstrated the superior outcomes of PRF when compared to PRP simply by removing anticoagulants from their formulations.2,9
Another drawback of PRP was the fact it remained liquid by nature (use of anticoagulants) and therefore when it was typically combined with biomaterials. When compared to PRF, PRP demonstrates a quick initial burst of growth factors being released, whereas PRF demonstrates a more slow and gradual release of growth factors over an extended period of time that has since been shown to significantly improve cell growth and tissue regeneration.10,11
Leukocyte and platelet-rich fibrin
Because of the main drawback that anticoagulants utilized in PRP prevented clotting, PRF was developed with the main aim of simply removing anticoagulants.12 By doing so, a much quicker working time was needed and the practitioner absolutely required centrifugation to begin shortly after blood collection (otherwise the blood would naturally clot within a tube). The main advantage of this fibrin matrix is the ability for it to release growth factors over an extended period of time while the fibrin clot is being degraded (as opposed to PRP, which remained in liquid form having a much faster growth factor release profile).13 Over the years, PRF has also been termed L-PRF (for leukocyte and platelet-rich fibrin) owing to the discovery that several leukocytes remained incorporated in PRF, which has been shown to increase antimicrobial properties14 as well as lead to less pain postoperatively.15
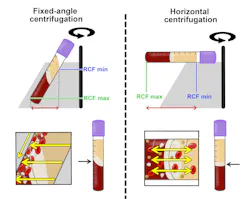
Recently, a series of basic laboratory experiments revealed better means to optimize the production of PRF using horizontal centrifugation. Simply, horizontal centrifuges are routinely utilized in high-end research labs as well as in medical hospitals due to their greater ability to separate layers based on density. Unlike a fixed-angle centrifugation system, whereby the tubes are actually inserted on an ~45-degree angle, horizontal centrifugation (often referred to as swing-out bucket centrifugation) enables the tubes to swing out to 90 degrees once they are in rotation (figure 1; QR code 2).
This technology leads to up to four times greater cell content when compared to fixed-angle centrifugation (figure 2).16
The major disadvantage of fixed-angle centrifugation is that during the spin cycles, cells are typically driven along the back wall of centrifugation tubes at high g-forces and are relatively difficult to separate properly according to their cell density. This also exposes cells to higher compressive forces against the back wall, and cells must then separate by travelling either up or down the inclined centrifugation slope based on their respective cell density differences. Since red blood cells (RBCs) are larger and heavier than platelets and leukocytes, they travel downward, whereas lighter platelets travel toward the top of the tube where PRF is collected. This makes it relatively difficult for the smaller cell types such as platelets and particularly leukocytes to reach the upper part of the tube, especially granted that RBCs outnumber white blood cells (WBCs) typically by ~1000 fold.
Therefore, and in summary, by utilizing a fixed-angle centrifuge, it is not possible to reach optimal accumulation of platelets or leukocytes as a result of their fixed-angle system design (QR code 3).
Dental uses of PRF
In dentistry, most procedures are done utilizing the 700 RCF for eight-minute protocol. Specifically to GBR, the so-called “sticky bone” protocol utilized this spin rate when both liquid-PRF (blue or white) and solid-PRF (red) tubes are drawn simultaneously and centrifuged at the same time (figure 3).
Always remember to draw liquid-PRF tubes first (QR code 4). While an entire textbook has been dedicated to this topic, briefly, PRF has shown effects for extraction site management, gingival recession coverage, and in implant dentistry to support soft tissue healing and reduce bacterial infiltration.1 Furthermore, PRF has been shown to speed healing in a wide variety of temporomandibular joint (TMJ) disorders17; its use has supported periodontal regeneration of intrabony18 and furcation defects,19 palatal wound healing,20 and has been utilized as a drug delivery system.21
Findings from preclinical trials and studies have found that 86% of all studies reported significant advantages when horizontal PRF was utilized compared to fixed-angle, with 14% of studies demonstrating no difference.22
Lastly, more recently, the use of a heating process to create denatured albumin gel (Alb-PRF) or extended-PRF (e-PRF)23 has demonstrated an ability to increase the resorption properties of regular PRF from a standard one- to two-week period to upward of four to six months.24-27
Also by the authors:
Editor’s note: This article originally appeared in Perio-Implant Advisory, a chairside resource for dentists and hygienists that focuses on periodontal- and implant-related issues. Read more articles and subscribe to the newsletter.
References
- Miron RJ, Estrin NE, Ahmad P, et al. Thirty years of autologous platelet concentrates: from platelet-rich plasma to platelet-rich fibrin. J Periodontal Res. 2025. Published online: August 4, 2025. doi:10.1111/jre.70013
- Miron RJ. Understanding Platelet Rich Fibrin. Quintessence; 2021.
- Anfossi G, Trovati M, Mularoni E, Massucco P, Calcamuggi G, Emanuelli G. Influence of propranolol on platelet aggregation and thromboxane B2 production from platelet-rich plasma and whole blood. Prostaglandins, Leukot Essent Fatty Acids. 1989;36(1):1-7. doi:10.1016/0952-3278(89)90154-3
- Fijnheer R, Pietersz RN, de Korte D, et al. Platelet activation during preparation of platelet concentrates: a comparison of the platelet-rich plasma and the buffy coat methods. Transfusion. 1990;30(7):634-638. doi:10.1046/j.1537-2995.1990.30790385523.x
- Upputuri PK, Sivasubramanian K, Mark CS, Pramanik M. Recent developments in vascular imaging techniques in tissue engineering and regenerative medicine. BioMed Res Int. 2015;2015:783983. doi:10.1155/2015/783983
- Gosain A, DiPietro LA. Aging and wound healing. World J Surg. 2004;28(3):321-326. doi:10.1007/s00268-003-7397-6
- Eming SA, Brachvogel B, Odorisio T, Koch M. Regulation of angiogenesis: wound healing as a model. Prog Histochem Cytochem. 2007;42(3):115-170. doi:10.1016/j.proghi.2007.06.001
- Eming SA, Kaufmann J, Lohrer R, Krieg T. [Chronic wounds. Novel approaches in research and therapy]. Hautarzt. 2007;58(11):939-944. doi:10.1007/s00105-007-1402-1
- Farshidfar N, Amiri MA, Estrin NE, et al. Platelet-rich plasma (PRP) versus injectable platelet-rich fibrin (i-PRF): a systematic review across all fields of medicine. Periodontol 2000. 2025. Published online: March 24, 2025. doi:10.1111/prd.12626
- Lucarelli E, Beretta R, Dozza B, et al. A recently developed bifacial platelet-rich fibrin matrix. Eur Cell Mater. 2010;20:13-23. doi:10.22203/ecm.v020a02
- Saluja H, Dehane V, Mahindra U. Platelet-rich fibrin: a second generation platelet concentrate and a new friend of oral and maxillofacial surgeons. Ann Maxillofac Surg. 2011;1(1):53-57. doi:10.4103/2231-0746.83158
- Choukroun J, Adda F, Schoeffler C, Vervelle A. Une opportunité en paro-implantologie: le PRF. Implantodontie. 2001;42(55):e62.
- Dohan Ehrenfest DM, Del Corso M, Diss A, Mouhyi J, Charrier JB. Three-dimensional architecture and cell composition of a Choukroun's platelet-rich fibrin clot and membrane. J Periodontol. 2010;81(4):546-555. doi:10.1902/jop.2009.090531
- Moraschini V, Miron RJ, Mourao CFdAB, et al. Antimicrobial effect of platelet‐rich fibrin: a systematic review of in vitro evidence‐based studies. Periodontol 2000. 2024;94(1):131-142. doi:10.1111/prd.12529
- Estrin NE, Tran TB, Ahmad P, et al. Analgesic effects of platelet-rich fibrin (PRF): a systematic review. Periodontol 2000. 2025. Published online: October 13, 2025. doi:10.1111/prd.70014
- Miron RJ, Chai J, Zheng S, Feng M, Sculean A, Zhang Y. A novel method for evaluating and quantifying cell types in platelet rich fibrin and an introduction to horizontal centrifugation. J Biomed Mater Res A. 2019;107(10):2257-2271. doi:10.1002/jbm.a.36734
- Farshidfar N, Amiri MA, Estrin NE, et al. Use of liquid platelet-rich fibrin (liquid PRF) in temporomandibular joint disorders: a narrative review with clinical recommendations. Periodontol 2000. 2025. Published online: September 26, 2025. doi:10.1111/prd.70012
- Miron RJ, Moraschini V, Estrin N, et al. Autogenous platelet concentrates for treatment of intrabony defects–a systematic review with meta-analysis. Periodontol 2000. 2025;97(1):153-190. doi:10.1111/prd.12598
- Miron RJ, Moraschini V, Estrin NE, et al. Periodontal regeneration using platelet-rich fibrin. furcation defects: a systematic review with meta-analysis. Periodontol 2000. 2025;97(1):191-214. doi:10.1111/prd.12583
- Farshidfar N, Ahmad P, Estrin N, Zhang Y, Miron RJ. Use of platelet-rich fibrin (PRF) on palatal wound healing: a narrative review with clinical recommendations. J Periodontal Res. 2025. Published online: June 10, 2025. doi:10.1111/jre.13413
- Miron RJ, Zhang Y. Autologous liquid platelet rich fibrin: a novel drug delivery system. Acta Biomater. 2018;75:35-51. doi:10.1016/j.actbio.2018.05.021
- Farshidfar N, Apaza Alccayhuaman KA, Estrin NE, et al. Advantages of horizontal centrifugation of platelet-rich fibrin in regenerative medicine and dentistry. Periodontol 2000. 2025. Published online: March 25, 2025. doi:10.1111/prd.12625
- Miron RJ, Pikos MA, Estrin NE, et al. Extended platelet-rich fibrin. Periodontol 2000. 2024;94(1):114-130. doi:10.1111/prd.12537
- Estrin NE, Ahmad P, Tran TB, et al. The evolution of extended platelet-rich fibrin membranes for socket grafting: part two: a randomized clinical trial comparing these membranes with collagen membranes. Dent J (Basel). 2026;14(1):45. doi:10.3390/dj14010045
- Estrin NE, Basma H, Espinoza AR, Pinto MAC, Pikos MA, Miron RJ. Extended platelet-rich fibrin as a membrane for lateral window sinus lifts: a case series. Clin Implant Dent Relat Res. 2025;27(1):e13427. doi:10.1111/cid.13427
- Estrin NE, Espinoza AR, Ahmad P, Imber JC, Farshidfar N, Miron RJ. The evolution of extended platelet-rich fibrin membranes for socket grafting: part one: technical development of protocols. Dent J (Basel). 2025;13(12):604. doi:10.3390/dj13120604
- Estrin NE, Tran TB, Espinoza AR, et al. Safety and feasibility of extended platelet-rich fibrin as a solo barrier membrane for ridge preservation: a case series. Clin Exp Dent Res. 2026;12(1):e70282. doi:10.1002/cre2.70282
About the Author

Richard Miron, DDS
Richard Miron, DDS, is currently lead educator and researcher at Advanced PRF Education and an adjunct visiting faculty in the department of periodontology at the University of Bern, Switzerland, where he completed his PhD studies since 2009. Dr. Miron is a prolific author and international lecturer on topics relating to growth factors, bone biomaterials, and guided bone regeneration. He is widely considered one of the top contributors to implant dentistry. He has written 10 textbooks on regenerative dentistry including Next Generation Biomaterials for Bone and Periodontal Regeneration, Understanding Platelet Rich Fibrin, and Modern Implant Dentistry.

Nathan Estrin, DMD
Nathan Estrin, DMD, received his bachelor’s degree in kinesiology from Indiana University and went on to earn his DMD from LECOM School of Dental Medicine. He received his training in periodontics at Stony Brook University in New York. During his dental studies, he developed a passion for regenerative, implant, and laser surgery and has more than 15 publications and three book chapters in these areas. Dr. Estrin is a board-certified periodontist in full-time private practice in Sarasota, Florida. He is also adjunctive faculty at LECOM School of Dental Medicine and a lead educator for PRFedu, where he trains dental offices on platelet-rich fibrin, lasers, and modern periodontal therapy.

Scott Froum, DDS
Editorial Director
Scott Froum, DDS, a graduate of the State University of New York, Stony Brook School of Dental Medicine, is a periodontist in private practice at 1110 2nd Avenue, Suite 305, New York City, New York. He is the editorial director of Perio-Implant Advisory and serves on the editorial advisory board of Dental Economics. Dr. Froum, a diplomate of both the American Academy of Periodontology and the American Academy of Osseointegration, is in the fellowship program at the American Academy of Anti-aging Medicine, and is a volunteer professor in the postgraduate periodontal program at SUNY Stony Brook School of Dental Medicine. He is a trained naturopath and is the scientific director of Meraki Integrative Functional Wellness Center. Contact him through his website at drscottfroum.com or (212) 751-8530.