The 2026 ACC/AHA dyslipidemia guidelines: What every dental professional needs to know
Key Highlights
- The 2026 ACC/AHA dyslipidemia guidelines introduce a new cardiovascular risk calculator (PREVENT-ASCVD) with lower LDL-C targets, signaling an era of earlier and more aggressive lipid-lowering intervention.
- Hyperlipidemia is associated with increased severity of periodontal disease, greater probing depths, clinical attachment loss, and elevated inflammatory mediators including IL-1β, TNF-α, and PGE2.
- Dyslipidemia has been identified as an independent risk factor for peri-implant bone loss and peri-implantitis, with meta-analytic data supporting this association.
- Statin medications—the pharmacologic cornerstone of dyslipidemia management—exhibit pleiotropic anti-inflammatory and bone-anabolic effects that may beneficially modulate periodontal and peri-implant tissue responses.
- Dental professionals are uniquely positioned to identify undertreated dyslipidemia and to integrate lipid status into periodontal risk stratification and implant candidacy assessment.
Introduction
On March 13, 2026, the American College of Cardiology (ACC) and the American Heart Association (AHA), alongside nine collaborating medical societies, released an updated guideline for the management of dyslipidemia—defined as abnormal levels of lipids or lipoproteins in the blood, including cholesterol and triglycerides. Published simultaneously in the Journal of the American College of Cardiology (JACC) and Circulation, this comprehensive document consolidates evidence-based recommendations for assessing and treating dyslipidemia to reduce an individual’s lifetime risk of atherosclerotic cardiovascular disease (ASCVD)—the leading cause of death globally, estimated to affect one in four US adults with elevated LDL-C.1
For the dental community, this guideline is not merely a cardiology update. Epidemiological and mechanistic evidence accumulated over the past two decades establishes a compelling, bidirectional relationship between dyslipidemia and oral health, specifically periodontal disease and peri-implant complications. As oral health professionals who interact with patients on a recurring basis, dentists and dental hygienists occupy a critical vantage point in the identification, monitoring, and comanagement of patients with hyperlipidemia. Understanding the 2026 guidelines—including their treatment targets, risk stratification tools, and pharmacologic recommendations—provides the dental team with a framework to better contextualize systemic disease burden in their patient population.
What’s new in the 2026 guidelines: A clinical summary2
The 2026 guideline introduces several clinically significant updates that depart meaningfully from prior recommendations:
- New risk calculator—PREVENT-ASCVD: Replacing the older Pooled Cohort Equations (PCE), which overestimated 10-year cardiovascular risk by 40%–50%, the new PREVENT-ASCVD equations estimate 10- and 30-year risk of heart attack or stroke in adults aged 30–79 without known ASCVD and with LDL-C between 70 and 189 mg/dL. Risk categories are reclassified as low (<3%), borderline (3% to <5%), intermediate (5% to <10%), and high (≥10%).
- Lower LDL-C targets: For primary prevention, LDL-C goals are now <100 mg/dL for borderline/intermediate risk and <70 mg/dL for high-risk patients. For secondary prevention in very high-risk ASCVD patients, the target drops to <55 mg/dL.
- Earlier pharmacologic intervention: If lifestyle modification fails to achieve target LDL-C, the guidelines recommend initiating lipid-lowering therapy earlier than previously considered, with statins remaining the pharmacologic foundation.
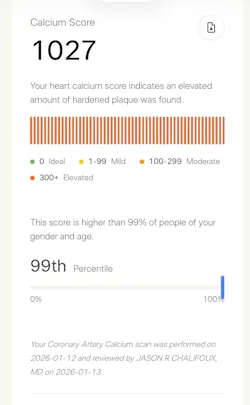
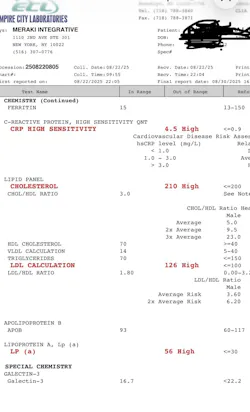
- Expanded biomarker assessment: Selective use of coronary artery calcium (CAC) score (figure 1), lipoprotein(a) [Lp(a)] measured at least once in adulthood, apolipoprotein B (apoB), and high-sensitivity C-reactive protein (hs-CRP) are recommended to refine individual cardiovascular risk profiles (figure 2).
- Risk enhancers including chronic inflammation: The guidelines explicitly recognize chronic inflammatory conditions as risk enhancers that modify ASCVD trajectory, a category directly relevant to periodontitis.
A critical nuance to the new aggressive targets: while intensive LDL-C lowering clearly reduces cardiovascular-specific mortality, a systematic review and meta-analysis by Ennezat et al. (2023) in the Journal of Cardiovascular Pharmacology found that the magnitude of all-cause mortality benefit does not scale linearly with depth of LDL-C reduction.3 Some data suggest a U-shaped relationship in which very low LDL-C levels (<50 mg/dL) may not confer additional survival benefit beyond cardiac event reduction. Large outcome trials such as FOURIER and ODYSSEY OUTCOMES have confirmed the safety of ultra-low LDL-C in high-risk populations, but the universal applicability of the <55 mg/dL target warrants individualized clinical judgment.4
Current pharmacologic therapies for dyslipidemia: What dental professionals should recognize
With a broadening pharmacologic armamentarium now codified in the 2026 guidelines, patients presenting to dental offices are increasingly likely to be on one or more lipid-lowering agents. A working familiarity with these medications—their mechanisms, systemic effects, and potential oral and osseous implications—is clinically relevant.
No. 1: Statins (HMG-CoA reductase inhibitors)
Statins remain the cornerstone of lipid-lowering pharmacotherapy. By inhibiting HMG-CoA reductase, they reduce hepatic cholesterol synthesis and upregulate LDL receptors, lowering circulating LDL-C by 30%–55% depending on agent and dose. Commonly prescribed agents include atorvastatin (Lipitor), rosuvastatin (Crestor), simvastatin (Zocor), and pravastatin. Beyond lipid lowering, statins exhibit significant pleiotropic effects including inhibition of NF-κB signaling, reduction of prostaglandin E2 production, downregulation of matrix metalloproteinases (MMPs), and stimulation of bone morphogenetic protein-2 (BMP-2). These mechanisms are directly relevant to periodontal and peri-implant biology. Some literature also suggests statins may offer bone-anabolic effects, potentially supporting alveolar bone density and osseointegration outcomes.5
No. 2: Ezetimibe (Zetia)
Ezetimibe inhibits intestinal cholesterol absorption via the NPC1L1 transporter and reduces LDL-C by an additional 15%–25% when added to statin therapy. The 2026 guidelines support its use when statin therapy alone is insufficient. No significant oral side effects are well documented, though its anti-inflammatory contribution is less robust than that of statins.
No. 3: Bempedoic acid (Nexletol)
A newer oral agent, bempedoic acid, inhibits ATP-citrate lyase (ACL), an enzyme upstream of HMG-CoA reductase in the cholesterol synthesis pathway. It offers an alternative for statin-intolerant patients, reducing LDL-C by approximately 15%–25%. The 2026 guidelines now explicitly recommend bempedoic acid as an adjunctive or alternative oral option. Early data suggest it also reduces hs-CRP, which is clinically significant given the shared inflammatory pathways between dyslipidemia and periodontal disease.
No. 4: PCSK9 monoclonal antibodies: evolocumab (Repatha) and alirocumab (Praluent)
PCSK9 inhibitors are the most potent LDL-C–lowering agents currently available, capable of reducing LDL-C by 50%–60% on top of statin therapy, achieving levels well below 55 mg/dL in high-risk patients. Administered subcutaneously every two to four weeks, they are recommended by the 2026 guidelines for patients with established ASCVD at very high risk who have not achieved LDL-C goals on oral therapy. Patients on PCSK9 inhibitors represent high cardiovascular risk profiles; dental clinicians should be attentive to coordinated perioperative planning in this population.
No. 5: Inclisiran (Leqvio)
Inclisiran is a small interfering RNA (siRNA) therapy that reduces hepatic PCSK9 production, achieving meaningful LDL-C reduction with twice-yearly injections following initial dosing. The 2026 guidelines note that outcome trial data are still accruing for inclisiran, but it represents an important adherence-friendly option for eligible patients.
No. 6: Fibrates and omega-3 fatty acids
Fibrates: fenofibrate (Tricor), gemfibrozil (Lopi), and prescription-strength omega-3 fatty acids (icosapentaenoic acid [EPA] and docosahexaenoic acid [DHA]) are primarily used to manage hypertriglyceridemia, a distinct therapeutic target highlighted in the 2026 guidelines. High-dose EPA (as in the REDUCE-IT trial) has demonstrated independent cardiovascular risk reduction. Of particular relevance to dental professionals: omega-3 fatty acids serve as precursors to pro-resolving lipid mediators, resolvins, and protectins that actively promote resolution of periodontal inflammation and may support tissue healing in the peri-implant environment.
Hyperlipidemia and periodontal disease: Mechanistic and clinical evidence
The relationship between hyperlipidemia and periodontal disease is bidirectional and mechanistically coherent. Both conditions share overlapping inflammatory pathways, and evidence suggests that dyslipidemia directly impairs periodontal tissue homeostasis. Fentoglu and Bozkurt (2008) provided an early framework for understanding this bidirectionality, demonstrating that periodontal infection elevates circulating lipopolysaccharide (LPS) from gram-negative periodontal pathogens, which stimulates hepatic production of very low-density lipoprotein (VLDL) and triglycerides while suppressing lipoprotein lipase activity, producing a net proatherogenic lipid profile.6 Conversely, hyperlipidemia promotes dysregulation of the host immune-inflammatory response to periodontal pathogens, amplifying tissue destruction.
Golpasand Hagh et al. (2014) demonstrated a statistically significant association between hyperlipidemia and periodontal infection severity, reporting elevated levels of IL-1β, TNF-α, and PGE2 in the gingival crevicular fluid of hyperlipidemic patients compared to normolipidemic controls.7 This pro-inflammatory cytokine milieu, identical to that which drives alveolar bone resorption in periodontitis, suggests that dyslipidemia amplifies the local inflammatory cascade triggered by periodontal pathogens, accelerating disease progression.
Awartani and Atassi (2010) further corroborated these findings in a direct clinical evaluation, identifying significantly greater probing depths, clinical attachment loss, and bleeding on probing in hyperlipidemic patients versus matched controls. Mechanistically, oxidized LDL (ox-LDL), a hallmark of dyslipidemia, activates macrophage foam cell formation, upregulates MMPs, and promotes endothelial dysfunction in periodontal microvasculature, collectively impairing tissue repair and regeneration.8 The resulting chronic inflammatory environment is permissive to deeper and more progressive periodontal destruction.
Hyperlipidemia and peri-implant disease: Emerging evidence
The implications of dyslipidemia extend beyond the natural dentition to the peri-implant environment. Gurgel et al. (2025), in a systematic review and meta-analysis published in the Journal of Clinical and Experimental Dentistry, specifically evaluated dyslipidemias as a risk factor for peri-implant complications. Their findings are clinically compelling: dyslipidemia was associated with statistically significant increases in peri-implant marginal bone loss and peri-implantitis incidence across included studies.9 The meta-analytic data support classifying dyslipidemia as an independent risk factor for peri-implant complications—a classification with direct implications for preoperative implant planning and long-term maintenance protocols.
The biological link is strong. Hyperlipidemia impairs osteoblast differentiation and function, elevates osteoclastic activity, reduces bone mineral density, and compromises osseointegration at the implant-bone interface.10 Elevated triglycerides and LDL-C alter the lipid microenvironment of bone marrow, promoting adipogenesis over osteogenesis and reducing the cellular substrate necessary for peri-implant bone regeneration.11 Additionally, the microvascular dysfunction associated with dyslipidemia may impair angiogenesis during the critical healing phase following implant placement. For implant surgeons and restorative clinicians, these findings necessitate a paradigm shift: dyslipidemic patients should be regarded with the same systemic scrutiny as diabetic or immunocompromised patients when assessing implant candidacy and designing peri-implant maintenance protocols.
Statins and the periodontium: A therapeutic intersection
An important and clinically actionable dimension of the dyslipidemia–oral health relationship is the potential therapeutic benefit of statin medications on periodontal and peri-implant tissues. Beyond lipid lowering, statins’ anti-inflammatory and bone-anabolic pleiotropic effects create a mechanistic rationale for improved oral health outcomes in patients on lipid-lowering pharmacotherapy. Several clinical studies have demonstrated reduced probing depths and improved clinical attachment levels in statin-using patients undergoing scaling and root planing.12 Local delivery of statins via biodegradable carriers has been explored as a site-specific adjunct to periodontal therapy with promising early results.
This therapeutic intersection also means that, as the 2026 guidelines drive an increasing proportion of at-risk patients toward earlier statin initiation, dental providers may observe a gradual improvement in the periodontal profile of their medically managed hyperlipidemic patients—provided oral hygiene and periodontal maintenance are optimized concurrently.
Clinical implications for dentists and dental hygienists
The 2026 ACC/AHA guidelines, integrated with the dental literature reviewed above, create a compelling framework for incorporating lipid status awareness into routine dental practice:
- Medical history review: Routinely document lipid panel values, dyslipidemia diagnoses, and current lipid-lowering medications. Patients on PCSK9 inhibitors or combination therapy represent high cardiovascular risk profiles and warrant coordinated care with their medical team.
- Periodontal risk stratification: Hyperlipidemia should be formally incorporated into periodontal risk assessments alongside diabetes, smoking, and genetic predisposition. Patients with uncontrolled dyslipidemia may warrant more frequent maintenance intervals and heightened clinical monitoring.
- Implant candidacy assessment: Preoperative lipid evaluation should be considered for patients planned for complex implant therapy, particularly those with borderline bone quality or a history of peri-implant complications. Optimizing dyslipidemia management prior to elective implant placement is prudent.
- Referral pathways: Dental professionals who identify patients with undiagnosed or poorly controlled dyslipidemia, evidenced by medical history, elevated hs-CRP, or a characteristic periodontal presentation, should refer proactively to the patient’s primary care physician or cardiologist.
- Patient education: The oral-systemic dialogue is a powerful motivational tool. Explaining that poor lipid control may worsen periodontal disease or compromise dental implant outcomes can meaningfully improve medical compliance in patients who are otherwise disengaged from cardiovascular risk management.
Conclusion
The 2026 ACC/AHA dyslipidemia guidelines represent a meaningful recalibration of cardiovascular risk management, one that extends its relevance well beyond the cardiology clinic. For dental professionals, this guideline is a direct call to action. The convergence of new LDL-C targets, expanded biomarker assessment, earlier pharmacologic intervention, and a growing body of evidence linking dyslipidemia to periodontal disease and peri-implant complications positions the dental team as an essential partner in systemic disease comanagement.
Hyperlipidemia amplifies periodontal inflammation, accelerates attachment loss, impairs osseointegration, and increases peri-implant bone loss. The pharmacologic agents now recommended for its treatment, particularly statins and emerging agents like PCSK9 inhibitors, may in turn offer ancillary oral health benefits through their anti-inflammatory and osseous effects. As the oral-systemic interface continues to be defined with greater precision, dental professionals who engage with lipid status as part of routine patient evaluation will be better positioned to deliver outcomes that reflect the true complexity of their patients’ overall health.
Editor’s note: This article originally appeared in Perio-Implant Advisory, a chairside resource for dentists and hygienists that focuses on periodontal- and implant-related issues. Read more articles and subscribe to the newsletter.
References
- 2025 heart disease and stroke statistics update fact sheet at-a-glance. American Heart Association. 2025. https://www.heart.org/en/-/media/PHD-Files-2/Science-News/2/2025-Heart-and-Stroke-Stat-Update/2025-Statistics-At-A-Glance.pdf
- Blumenthal R, Morris PB, Gaudino M, et al. 2026 ACC/AHA/AACVPR/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Dyslipidemia: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2026:S0735-S1097(25)10254-4. doi:10.1016/j.jacc.2025.11.016
- Ennezat PV, Guerbaai RA, Maréchaux S, Le Jemtel TH, François P. Extent of low-density lipoprotein cholesterol reduction and all-cause and cardiovascular mortality benefit: a systematic review and meta-analysis. J Cardiovasc Pharmacol. 2023;81(1):35-44. doi:10.1097/FJC.0000000000001345
- Rabih AM, Niaj A, Raman A, et al. Reduction of cardiovascular risk using proprotein convertase subtilisin/kexin type 9 inhibitors in patients with acute coronary syndrome: a systematic review. Cureus. 2023;15(2):e34648. doi:10.7759/cureus.34648. Erratum in: Cureus. 2025;17(8):c270. doi:10.7759/cureus.c270
- Kabra S, Thosar NR, Malviya NS. Exploring the synergistic effect of simvastatin in oral health applications: a literature review. Cureus. 2023;15(8):e44411. doi:10.7759/cureus.44411
- Fentoglu O, Bozkurt FY. The bi-directional relationship between periodontal disease and hyperlipidemia. Eur J Dent. 2008;2(2):142-146.
- Golpasand Hagh L, Zakavi F, Hajizadeh F, Saleki M. The association between hyperlipidemia and periodontal infection. Iran Red Crescent Med J. 2014;16(12):e6577. doi:10.5812/ircmj.6577
- Awartani F, Atassi F. Evaluation of periodontal status in subjects with hyperlipidemia. J Contemp Dent Pract. 2010;11(2):033-040.
- Gurgel B, Souza R, Vajgel A, Cabral MF, Almeida M, Almeida R. Are dyslipidemias a risk factor for peri-implant complications? A systematic review and meta-analysis. J Clin Exp Dent. 2025;17(4):e461-e473. doi:10.4317/jced.62241
- Tintut Y, Morony S, Demer LL. Hyperlipidemia promotes osteoclastic potential of bone marrow cells ex vivo. Arterioscler Thromb Vasc Biol. 2004;24(2):e6-e10. doi:10.1161/01.ATV.0000112023.62695.7f
- Pappan N, Awosika AO, Rehman A. Dyslipidemia. In: StatPearls [Internet]. StatPearls Publishing; 2026. https://www.ncbi.nlm.nih.gov/books/NBK560891/
- Cecoro G, Piccirillo A, Martuscelli G, Del Fabbro M, Annunziata M, Guida L. Efficacy of locally delivered statins as an adjunct to scaling and root planning in the treatment of periodontitis: a systematic review and meta-analysis. Eur Rev Med Pharmacol Sci. 2021;25(18):5737-5754. doi:10.26355/eurrev_202109_26792
About the Author

Scott Froum, DDS
Editorial Director
Scott Froum, DDS, a graduate of the State University of New York, Stony Brook School of Dental Medicine, is a periodontist in private practice at 1110 2nd Avenue, Suite 305, New York City, New York. He is the editorial director of Perio-Implant Advisory and serves on the editorial advisory board of Dental Economics. Dr. Froum, a diplomate of both the American Academy of Periodontology and the American Academy of Osseointegration, is in the fellowship program at the American Academy of Anti-aging Medicine, and is a volunteer professor in the postgraduate periodontal program at SUNY Stony Brook School of Dental Medicine. He is a trained naturopath and is the scientific director of Meraki Integrative Functional Wellness Center. Contact him through his website at drscottfroum.com or (212) 751-8530.