Creating keratinized soft tissue to enhance the implant restoration
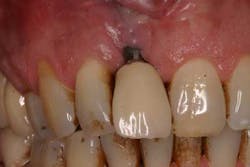
Fig. 1: Soft tissue recession around a dental implant restoration due to a deficiency in soft tissue volume.
There are a number of surgical and prosthetic techniques used to preserve existing peri-implant soft tissue prior to or during implant surgery. Examples include papilla sparring incisions (7), atraumatic tooth extraction, immediate implant placement with or without immediate provisionalization (8-12), and platform switching (13). These techniques are predicated on the fact that sufficient soft tissue exists in order to create an esthetic gingival-restorative complex.
A problem arises when there is a paucity of soft tissue to develop during an implant restoration. Although there is a controversy as to whether an “adequate” band of keratinized tissue is needed to achieve a long-term esthetic and functional restorative result (14-16), most authors would agree that having keratinized tissue around implant restorations can only be beneficial. Because the attachment apparatus is different between a tooth and a dental implant (17), in conjunction with a more limited blood supply to an implant compared with a tooth (18), implants are typically more susceptible to breakdown (19).
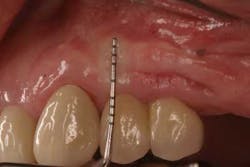
Keratinized tissue around implants can be one method of increasing resistance to bacterial infection. In fact, in a recent review by Greenstein and Cavallaro, the authors listed several indications that necessitated keratinized tissue presence around dental implants. These included patients with chronic inflammation despite hygiene efforts (Fig. 2), continued recession or attachment loss despite periodontal intervention, sites with soreness upon brushing, a predisposition toward periodontitis or recession, noncompliant patients, and those patients who want to improve esthetics (20).
Fig. 2: Persistent inflammation around a dental implant restoration with deficient keratinized tissue despite adequate oral hygiene.
Several surgical techniques have been described in the literature to increase the amount of keratinized tissue around teeth and dental implants. They include autogenous soft tissue grafts from the patient such as the free gingival graft or the subepithelial connective tissue graft, allogenic soft tissue grafts from human cadavers, xenogenic soft tissue grafts from animals, and newer cell-engineered grafts in culture dishes. Although autogenous tissue grafting is considered the gold standard in soft tissue augmentation procedures, much attention has been given to alternative procedures due to the morbidity associated with harvesting autografts.
Mucograft® is an example of a xenograft material that has been used in lieu of autogenous grafts to increase keratinized tissue around dental implants (21). Made of porcine collagen, this material has a compact outer layer that can be left exposed to the oral environment and a spongy inner layer that facilitates a blood clot and promotes angiogenesis. A major advantage in using this material for increasing keratinized tissue around dental implants is that it can be used in challenging areas with little to no attached soft tissue because primary closure over this graft material is not needed and it can be left exposed (Figs. 3a-3c).
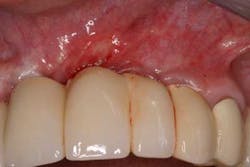
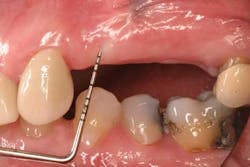
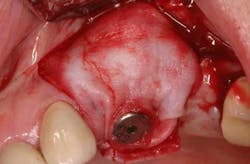
Fig. 3a: Edentulous Nos. 12 and 13 slated for implant therapy with a limited amount of keratinized tissue.
Fig. 3b: Soft tissue Mucograft placed at the time of implant surgery in order to augment keratinized tissue height and thickness. Primary closure was not obtained over this graft.
Fig. 3c: Final restorations showing a large increase in the height of keratinized tissue, facilitating oral hygiene and increasing plaque resistance.
Editor’s note: Drs. Scott Froum and Chris Salierno will be presenting on implant complications on Aug. 4, 2012, in Las Vegas. For more information, click here.
Author bio
Scott Froum, DDS, is a periodontist and co-editor of Surgical-Restorative Resource e-newsletter. He is a clinical associate professor at the New York University Dental School in the Department of Periodontology and Implantology. He is in private practice in New York City. You may contact him through his website at www.drscottfroum.com.
References
1. Branemark Pl, Hansson BO, Adell R, et al. Osseointegrated implants in the treatment of the edentulous jaw. Experience from a ten-year period. Scand J Plast Reconstr Surg 1977; 16 (Suppl):1-132.
2. Adell R, Lekholm U, Rockler B, et al. A 15-year study of osseointegrated implants in the treatment of the edentulous jaw. Int J Oral Surg 1981; 10:387-416.
3. Albrektson T. A multicenter report on osseointegrated oral implants. J Prosthet Dent 1988; 60:75-84.
4. Lazzara R, Siddiqui AA, Binon P, et al. Retrospective multicenter analysis of 3i endosseous dental implants placed over a five-year period. Clin Oral Implants Res 1996; 7:73-83.
5. Lekholm U, Gunne J, Henry P, et al. Survival of the Branemark implant in partially edentulous jaws: A 10-year prospective multicenter study. Int J Oral Maxillofac Impl 1999; 14:639-645.
6. Buser D, Mericske-Stern R, Bernard JP, et al. Long-term evaluation of non-submerged ITI Implants. Part 1: 8-year life table analysis of a prospective multi-center study with 2,359 implants. Clin Oral Implants Res 1997; 8:161-172.
7. Joseph Y Kan, Kitichai. K. Rungcharassaeng: Interimplant papilla preservation in the esthetic zone: a report of six consecutive cases. Int J Periodontics Restorative Dent 2003; 23:249–259.
8. Froum SJ. Immediate placement of implants into extraction sockets: rationale, outcomes, technique. 2005; 98:19-33.
9. Rosenquist B, Grenthe B. Immediate placement of implants into extraction sockets: implant survival. Int J Oral Maxillofac Implants 1996; 11:205-209.
10. Kan J, Rungcharassaeng K, Lozada J. Immediate placement and provisionalization of maxillary anterior single implants: 1-year prospective study. Int J Oral Maxillofac Implants 2003; 18:31-39.
11. De Kok I, Chang SS, Moriarty JD, Cooper LF. A retrospective analysis of peri-implant tissue responses at immediate load/provisionalized microthreaded implants. Int J Oral Maxillofac Implants 2006; 21:405-412.
12. Covani U, Barone A, Cornellini R, Crespi R. Soft tissue healing around implants placed immediately after tooth extraction without incision: a clinical report. Int J Oral Maxillofac Implants 2004; 19:549-553.
13. Lazzara RJ, Porter SS. Platform switching: a new concept in implant dentistry for controlling postoperative crestal bone levels. Int J Peri Rest Dent 2006; 26:9-17.
14. Wennström JL, Bengazi F, Lekholm U. The influence of the masticatory mucosa on the peri-implant soft tissue condition. Clin Oral Implants Res. 1994; 5(1):1-8.
15. Buser D, Mericske-Stern R, Bernard JP, et al. Long-term evaluation of nonsubmerged ITI implants. Part 1: 8-year life table analysis of a prospective multi-center study with 2359 implants. Clin Oral Implants Res. 1997; 8(3):161-172.
16. Chung DM, Oh TJ, Shotwell JL, et al. Significance of keratinized mucosa in maintenance of dental implants with different surfaces. J Periodontol. 2006; 77(8):1410-1420.
17. Berglundh T, Lindhe J, Ericsson I, et al. The soft tissue barrier at implants and teeth. Clin Oral Implants Res. 1991; 2(2):81-90.
18. Berglundh T, Lindhe J, Jonsson K, Ericsson I. The topography of the vascular systems in the periodontal and peri-implant tissues in the dog. J Clin Periodontol. 1994; 21(3):189-193.
19. Lindhe J, Berglundh T, Ericsson I, et al. Experimental breakdown of peri-implant and periodontal tissues. A study in the beagle dog. Clin Oral Implants Res. 1992; 3(1):9-16.
20. Greensteen G, Cavallaro J. The clinical signifigance of keratinized tissue around dental implants. Compend Contin Educ Dent. October 2011; 32(8):24-31.
21. Sanz M, Lorenzo R, Aranda JJ, Martin C, Orsini M. Clinical evaluation of a new collagen matrix (Mucograft prototype) to enhance the width of keratinized tissue in patients with fixed prosthetic restorations: a randomized prospective clinical trial. J Clin Periodontol 2009; 36: 868-876.