Addressing the connection leakage issues in the dental implant/abutment space
The material you choose to close the screw access chamber with screw-retained implants can be an important factor when considering leakage and bacterial contamination of the dental implant/abutment. Leakage, which can impact the success and longevity of dental implants, has been identified as a major contributing factor to the introduction of bacteria and inflammatory reactions at the implant-abutment connection, leading to problems such as oral malodor, inflammation, and peri-implant bone loss (figure 1).
Peri-implant mucositis and peri-implantitis are the primary diseases affecting dental implant health. Peri-implantitis is a pathological condition occurring in tissues around dental implants, characterized by inflammation in the peri-implant connective tissue and progressive loss of supporting bone.
How microgaps affect dental implant success
One of the causes of peri-implantitis can be attributed to microgaps, or inadequate sealing around the implant, that can allow bacteria to infiltrate, leading to inflammation and infection of the peri-implant tissues. If left untreated, peri-implantitis can cause bone loss, implant instability, and, ultimately, implant failure.
Studies have demonstrated that during mastication, fluid can enter microgaps in the implant/abutment space, allowing bacteria and endotoxins to enter.1 This microleakage is an important risk factor for complications such as bone resorption, peri-implant inflammation, and central screw loosening.2
Filling the screw access chamber
Commonly utilized materials to fill the screw access chamber—including cotton pellets, polyethylene tape, rubber O-rings, and metal caps—are known to be nonhygienic, absorb moisture, harbor bacteria, and prove challenging to remove. Many have experienced unpleasant smells when accessing the screw space covered by these materials.
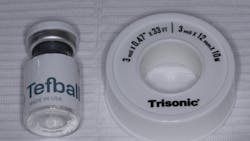
Tefball serves the same purpose as materials like cotton pellets, PVS, and Teflon tape, acting as an interface between the screw hole of a dental implant and the composite filling cover. The superiority of this product lies in its antimicrobial properties as well as its unique physical design, material composition, and durability (figure 2).
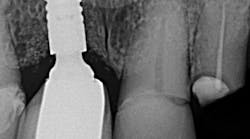
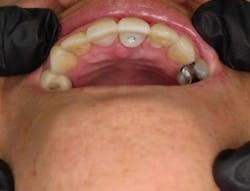
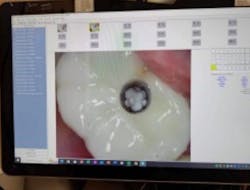
Tefball is perfectly sized to fit implant access holes, featuring a distinctive design crafted to prevent composite infiltration while ensuring optimal engagement for effective retention (figure 3). It is explicitly calibrated to match the diameter of the access cavity, providing a seamless and efficient solution for your dental implant needs (figure 4). Using Tefball saves time and money and ensures a better outcome for patients. The product stands out for its cleanliness, antimicrobial properties, hygiene, and ease of insertion and removal.
Preventing microgap leakage
Material choice should be based on the specific requirements of each implant case, the dentist’s preferences, and compatibility with the implant system used. Additionally, precise clinical techniques and attention to detail during placement are essential for an effective seal that prevents abutment connection leakage. Regular follow-up and maintenance should be integral parts of the overall treatment plan to monitor the sealing material’s integrity and promptly address any issues that may arise.
Editor’s note: This article originally appeared in Perio-Implant Advisory, a chairside resource for dentists and hygienists that focuses on periodontal- and implant-related issues. Read more articles and subscribe to the newsletter.
References
- Sahin C, Ayyildiz S. Correlation between microleakage and screw loosening at implant-abutment connection. J Adv Prosthodont. 2014;6(1):35-38. doi:10.4047/jap.2014.6.1.35
- Jervøe-Storm PM, Jepsen S, Jöhren P, Mericske-Stern R, Enkling N. Internal bacterial colonization of implants: association with peri-implant bone loss. Clin Oral Implants Res. 2015;26(8):957-963. doi:10.1111/clr.12421
Michael Lowe, DDS, completed his undergraduate degree at Michigan State University and his Doctor of Dental Surgery degree at Dalhousie University in Halifax, Nova Scotia. He is a recipient of the prestigious Canadian Dental Association President’s Award and the American College of Dentists’ Award. He is an active member of the Canadian Dental Association, British Columbia Dental Association, and Nova Scotia Dental Association. He maintains his license to practice dentistry in Oregon as well as in Canada. Dr. Lowe is the owner of the company that manufactures Tefball.
About the Author

Michael Lowe, DDS
Michael Lowe, DDS, completed his undergraduate degree at Michigan State University and his Doctor of Dental Surgery degree at Dalhousie University in Halifax, Nova Scotia. He is a recipient of the prestigious Canadian Dental Association President’s Award and the American College of Dentists’ Award. He is an active member of the Canadian Dental Association, British Columbia Dental Association, and Nova Scotia Dental Association. He maintains his license to practice dentistry in Oregon as well as in Canada. Dr. Lowe is the owner of the company that manufactures Tefball.
Updated March 12, 2024